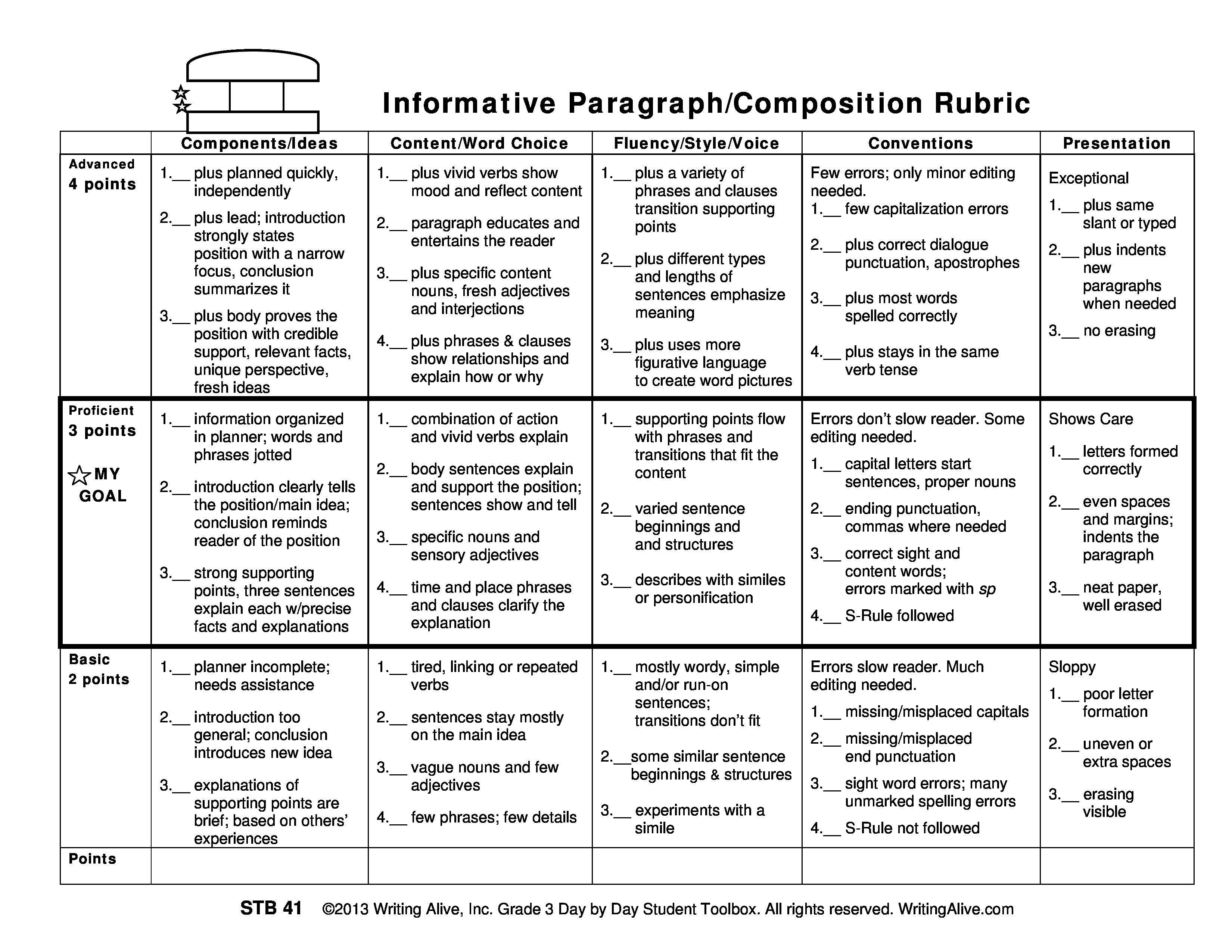
What is the chemical equation for the combustion of propane.
Write A Balanced Equation For The Combustion Of Propane C3h8, essays on why not to pay taxes, cover letter resources, essay on whale rider.
Home Propane (C3H8) Propane (C3H8) Propane (C 3 H 8) Propane is a three-carbon alkane, normally a gas, but compressible to a transportable liquid. It is derived from other petroleum products during oil ornatural gas processing. It is commonly used as a fuel for engines, oxy-gas torches, barbecues, portable stoves and residential central heating. When used as vehicle fuel, it is commonly known.
Write a balanced equation for the combustion of propane C3H8 When balanced the from SCIENCE 100 at Ocean View High School.
Propane (c3h8) burns in oxygen to produce carbon dioxide gas and water vapor. (a) write a balanced equation for this reaction. (use the lowest possible coefficients. include states-of-matter under the given conditions in your answer.).
I need help with this problem: Propane gas (C3H8) burns completely in the presence of oxygen gas (O2) to yield carbon dioxide gas (CO2) and water vapor (H2O). 1. Write a balanced equation for this reaction. 2. Assuming that all volume measurements occur at the same temperature and pressure, how many liters of oxygen will be required to completely burn 0.700 L of propane gas?
Answer (1 of 1): This equation is known as the Oxidation of Propane. In the equation, C3H8 is propane, which is going through the process of oxidation. After reaction carbon dioxide and water is producing. When organic compounds are burnt then carbon dioxide and water is produced.
Propane, C3H8, is a hydrocarbon that is commonly used as fuel for cooking. (a) Write a balanced equation for the complete combustion of propane gas, which yields CO2(g) and H2O(l). (b) Calculate the volume of air at 30 C and 1.00 atmosphere that is needed to burn completely 10.0 grams of propane. Assume that air is 21.0 percent O2 by volume. (c) The heat of combustion of propane is -2,220.1 kJ.